We had a UsefulChem group meeting yesterday Oct 2, 2006 with James, Khalid, Alicia, Dave and a new member Sean.
On the laboratory front, we finally have access to both C and H NMR. With the synthesis of
DOPAL solved, the obstacle to getting our anti-malarial compounds made remains carrying out a
Ugi reaction followed deprotection then cyclization (UDC). We have been following a
paper with limited experimental data which has made it difficult to tell where things are failing. It would be helpful to replicate a prior published example of a UDC strategy to generate a diketopiperazine and compare NMRs to make sure that we can at least do that. So far we have not found such a paper or report but here is a place where we are
collecting references. Please add relevant links.
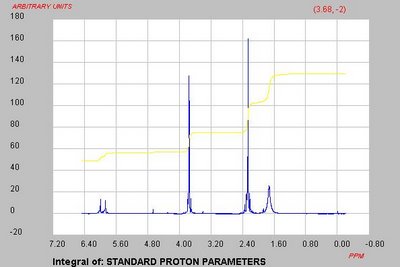
So I think we need to make a better effort to make, isolate and characterize a Ugi product and then worry about the deprotection and cyclization. Having access to C NMR is going to make this much easier. In fact Lin is analyzing a
simple Ugi reaction with piperonal so we may be close to getting a handle on this first step.
I spent some time going over
Armstrong's paper on the convertible isonitrile in the Ugi reaction. Every lab member working on this project needs to read this paper carefully. There are some detailed experimental procedures for carrying it out the Ugi reaction and some discussion about the mechanism. Note especially that the proposed mechanism on p. 2577 shows a protonated imine serving as a leaving group to drive the reaction. I don't think a protonated amine, which we would have during the cyclization step using our benzyl isonitrile derived Ugi products, should behave that differently. I'm willing to accept that it might but I don't know why. Any ideas? Unfortunately Armstrong did not test another isonitrile in this paper and he did not make diketopiperazines, although he does demonstrate cyclizations to seven membered rings.
In case we do need the
cyclohexenyl isonitrile used by Armstrong, Khalid has located a company and is awaiting pricing info. When we looked
previously it was not commercially available.
Otherwise Dave talked about his progress on the
cheminformatics front and Sean had to leave before reporting on his
docking calculations. More on this later.